Production of Penicillin
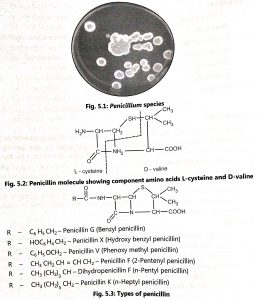
Production of penicillin began in the United States (1941) by surface culture fermentation of Penicillium notatum. During World War II, penicillin-producing fungi were studied extensively to increase yields of penicillin. Now a day, penicillin is produced from Penicillium chrysogenum by submerged culture techniques. The morphology of Penicillium species is shown in Fig. 5.1. Penicillin is effective against Gram-positive bacteria and also some large viruses and Rickettsia. Penicillin is a generic term applied to an entire group of antibiotics.
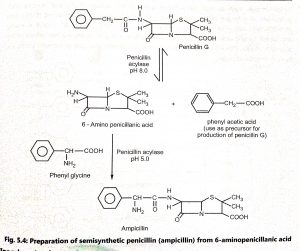
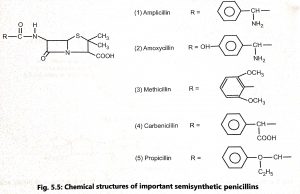
The penicillin nucleus contains two amino acids, L-cysteine and D-valine (Fig. 5.2) which are closely related in structure. Penicillins are N-acyl derivatives of 6-aminopenicillanic acid (6-APA). By adding side chains to the penicillin nucleus, new penicillins are synthesized (Fig. 5.3). The semisynthetic penicillins are prepared from 6-aminopenicillanic acid (6-APA). Acylation of the 6-APA is easily and efficiently brought about by chemical reactions (Fig. 5.4) Different derivatives of semisynthetic penicillin are shown in Fig. 5.5.
Inoculum development:
The selected strain of Penicillium chrysogenum is maintained in the form of a master culture and preserved by lyophilization, or by fixing the spores in sterilized soils and mixtures. For inoculum preparation, spores from working solid cultures are suspended in water. These spores are added in flasks containing nutrient solution and incubated for 4 to 6 days at 25°C. The resulting spores are used directly to inoculate inoculum tank. The inoculum tanks are incubated for 48 hours with agitation and aeration to grow more mycelium. The resulting inoculum is used for a production tank or it is added to a second or even third-stage inoculum tank to produce more inoculum for large-scale fementation.
Production media:
The exact composition of penicillin production media used in the industry for production of penicillin is unknown. These media are considered to be trade secrets of that particular fermentation industry. A typical medium described by Jackson (1958) contains fermentable carbohydrates, such as corn steep liquor solids (3.5%), lactose (3.5%) and glucose (1%) potassium dihydrogen phosphate (0.4 %), calcium carbonate (1%), edible oil (04%), and penicillin precursor. The pH of this medium after sterilization is 5.5 to 6.0. Inoculum media are similar to production media except that lactose and precursors are not added in the inoculum media. These media compositions may be slightly changed to increase yields and meet economic changes.
Fermentation:
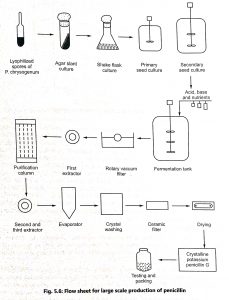
The media are placed in a fermentation vessel, sterilized, and inoculated with a suspension of Penicillium chrysogenum (inoculum). A flowsheet for large-scale production of penicillin is shown in Fig. 5.6. The fermentation vessel is equipped with devices that allow continuous addition of nutrients, acids/bases to maintain the pH (7 to 7.4), and cooling coils to maintain the temperature (24°C).
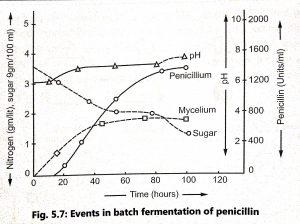
The mycelium grows rapidly by utilizing organic nitrogen compounds and lactic acid as a source of carbon. Ammonia is liberated by deamination of the amino acids of the corn steep liquor and pH rises. Ammonia is actively assimilated and maintains the pH. Active penicillin production is associated with this phase of lactose and ammonia utilization (Fig. 5.7). Maximum antibiotics are produced within 4 to 5 days.
Recovery and purification:
A schematic flow diagram for the recovery of potassium penicillin G (down stream process) is incorporated in Fig. 5.6. The first step in the recovery process is the removal of mycelium or cells by filtration (rotary vacuum filter) or centrifugation. These stages are carried out under aseptic conditions, to avoid contamination of the filtrate with penicillinase-producing microorganisms (Bacillus species) which may cause loss of antibiotics. Penicillin is extracted under controlled conditions of temperature, pH and sterility to minimize chemical and enzymatic degradation. It is extracted in the form of acid into amyl acetate, methyl isobutyl ketone, or butyl acetate in a counter-current solvent extractor at pH 2.5 to 3.0.
A penicillin-containing solvent is treated with active charcoal to remove pigments and other impurities. The charcoal is separated from the extract on a precoated rotary vacuum filter and then washed with the solvent. Penicillin from the solvent is crystallized by the addition of sodium or potassium hydroxide to form its salt. The end product of penicillin is then crystallized into sodium or potassium penicillin.
