NON-RADIOACTIVE HYBRIDIZATION PROCEDURES
Nucleic acid hybridization can be detected by labeling the probe with a radioactive isotope or a non-radioactive isotope. Therefore, the hybridization procedure can be of two types depending upon the type of label used to label the probe.
Radioactive Hybridization Procedure
Radioactive hybridization procedures are used in most of the research laboratories. Nucleotides in this method are labeled with a radioactive isotope and thus resulting in the formation of labelled nucleic acids. These radioactively labeled nucleic acids are then used as probe in radioactive hybridization procedures. In a standard radioactive hybridization detection system, the sample DNA is first bound to a solid support. Then, radioactively labeled probe is mixed with the sample DNA. Then a washing is given to remove any non-hybridized probe from the mixture. The radio-labeled probes are then detected (in solution or within a solid sample) by autoradiography. The most commonly used radioactive isotopes are 32P, 33P, 3H and 7S. Amongst these radio-isotopes 32P is most widely used because 32P emits β-particles (high energy) which have a high degree of sensitivity for detection. But 32P has many disadvantages also. Such as:
- Short lived
- Dangerous
- Specific laboratory equipments are required for handling and disposal.
Therefore, non-radioactive hybridization procedures have been developed.
Non-Radioactive Hybridization Procedure
Non-radioactive hybridization procedures have widespread applications in different areas. The non-radioactive hybridization procedures amplifies a signal (detects the DNA sample) by enzymatic conversion of chromogenic or chemilumniscent substrates. Chromogenic substrates changes color and chemiluminescent substrates produces light after conversion into a specific product by a suitable enzyme. There are two types of non-radioactive labeling procedures:
- Direct non-radioactive labelling
- Indirect non-radioactive labeling
(a) Direct non-radioactive labeling – In direct non-radioactive labeling, a labeled group is attached to a nucleotide and then this labeled nucleotide is incorporated in the reaction mixture. Labeling group in this method can be a fluorophore (chemical group which fluoresce when exposed to light of certain wavelength). Examples of fluorophores are fluorescein (green), rhodamine (red), texas red (red), AMCA (blue), CY3 (red), etc.
(b) Indirect non-radioactive labeling — In indirect non-radioactive labeling (Fig. 25.1), a reporter molecule (R) is coupled (chemically) to the nucleotide. This reporter labeled nucleotide then gets incorporated into DNA. An affinity molecule (A) which has very high specificity for the reporter molecule is initially conjugated to a marker molecule (M, detected with the help of an assay) which can be detected and then added to the reaction mixture. The affinity molecule will find to reporter molecule only and the unbound molecules are removed away by giving a washing. The molecule which has high affinity for the reporter molecule is often a specific antibody or a ligand. The marker molecule (M) can be detected by many assays. Such as, if it labeled with a specific fluorescent dye, it is detected by fluorometric assay; if it is labelled with an enzyme, it can be detected by an enzyme assay which can then be measured colorimetrically.
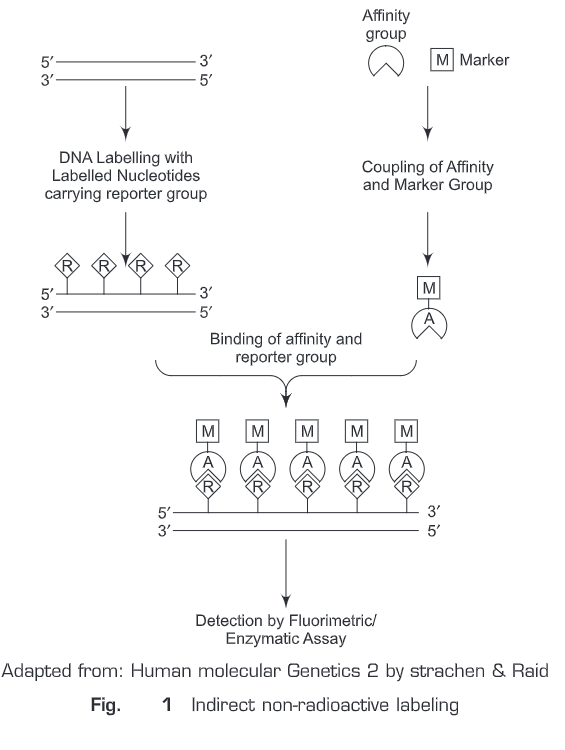
Two widely used indirect non-radioactive labeling sytems are:
- Biotin streptavidin system (Fig. 25.2)— In this system , two ligands are used
(i) Biotin (a natural vitamin)
(ii) Streptavidin (bacterial protein) or avidin (chicken egg white protein)
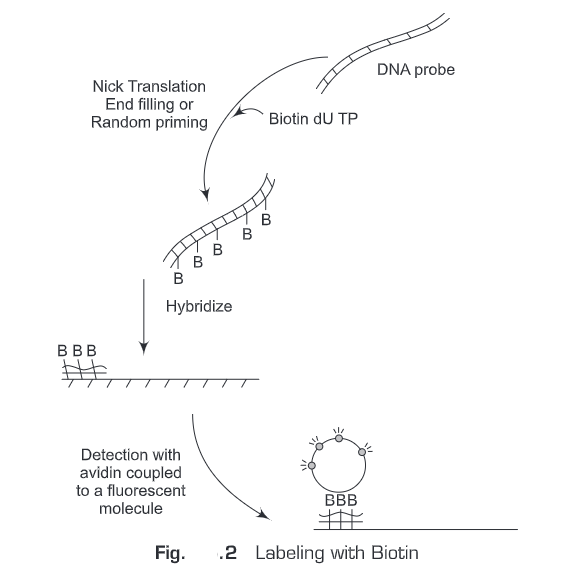
Biotin works as reporter molecule. Streptavidin has very high affinity for biotin molecule (affinity constant = 10-14). The standard procedure involves the following steps:
- Biotin is conjugated to nucleotides
- Biotin-labeled nucleotide (probe) is hybridized to the target DNA
- Streptavidin is added.
- Alkaline phosphatase (AP) conjugated to biotin (biotin-labeled enzyme) is added
- Then a substrate is added
- The product is then measured by enzymatic assay \ depending upon the type of the substrate used
Streptavidin and avidin both have four biotin binding sites. Therefore, a single streptavidin or avidin molecule can bind to biotin-labeled probe and biotin labeled enzyme simultaneously. The activity of the enzyme is not hampered by streptavidin/avidin binding or biotin labeling.
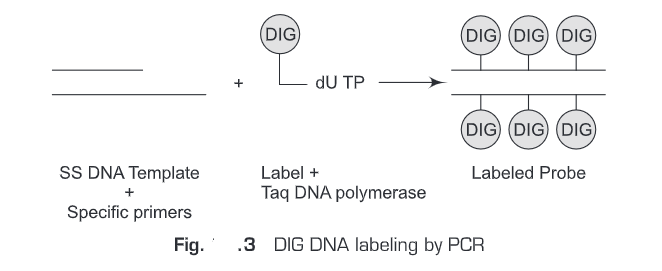
- Digoxigenin system- Digoxigenin is a plant steroid obtained from Digitalis. A specific antibody is raised against digoxigenin. This Digoxigenin-specific antibody is then used for the detection. Digoxigenin works as a reporter molecule and Digoxigenin-specific antibody as affinity molecule. The standard procedure involves the following steps (Fig. 25.3).
- Digoxigenin is conjugated to nucleotides.
- Digoxigenin-labeled nucleotide (probe) is hybridized to the target DNA.
- Digoxigenin-specific antibody is added.
- Alkaline phosphatase (AP) conjugated to Digoxigenin (Digoxigenin-labeled enzyme) is added.
- Then a substrate is added.
- The product is then measured by enzymatic assay depending upon the type of the substrate used.
Non-Radioactive hybridization procedures can also be classified into two other classes depending upon the type of substrate used for signal amplification. These are :
(a) Chromogenic detection systems— Chromogenic substrate changes colour after conversion into a specific product by a suitable enzyme which is then measured colorimetrically.
(b) Chemiluminesent detection systems (Fig. 25.4)- Chemiluminescent substrates produces light after conversion into a specific product by a suitable enzyme which is then measured by luminometer.
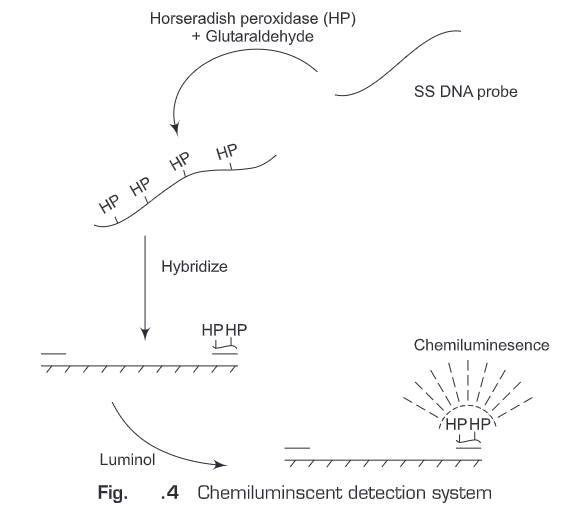
Non-Radioactive hybridization procedures have many more advantages. Such as:
- Detection can be completed within few hours.
- Biotin labeled probe is stable for one year at room temperature.
- Chemiluminesence is highly sensitive method.
Questions
1. What are the commonly used radioactive isotopes in hybridization?
2. Which particle 32P emits?
3. What are the advantages of 32P?
4, What are the different methods for labeling of probes?
5. What are the advantages of non-radioactive labeling procedure for nucleic acid hybridization over radioactive procedures?
6. What are chromogenic and chemiluminescent substrates?
7. What are the different non-radioactive labeling procedures?
8. What is a fluorophore? Give examples.
9. Explain the indirect non-radioactive labeling method.
10. What is avidin, biotin and streptavidin?
11. How many binding sites do avidin and streptavidin have?
12. What is digoxigenin?
13. What are the differences between chromogenic and chemiluminescent detection systems?
14. Write a short note on molecular beacon.
