Introduction
Methods of bacterial culture used in the clinical laboratory include streak culture, lawn culture, stroke culture, stab culture, pour-plate culture, shake culture, and liquid culture. Special methods are employed for culturing anaerobic bacteria. The sweep plate method is used for estimating bacteria in the dust on clothing.
Streak Culture (Surface Plating)
- This method is routinely employed for the isolation of bacteria in pure culture from clinical specimens.
- A platinum loop is charged with the specimen to be cultured. Owing to the high cost of platinum, loops for routine work are made of nichrome resistance wire.
- The loop is flat, circular, and completely closed with a 2-4 mm internal diameter mounted on a handle. One loopful of the specimen is smeared thoroughly over an area, on the surface of a well-dried plate, to give a well-inoculum or ‘well’.
- The loop is re-sterilized and drawn from the well in two or three parallel lines onto the fresh surface of the medium. This process is repeated several times.
- At each step, the inoculum is derived from the most distal part of the immediately preceding strokes.
- Plates are incubated in the inverted position with the lid underneath. On incubation, growth may be confluent at the site of original inoculation (well) but becomes progressively thinner, and well-separated colonies are obtained over the final series of streaks
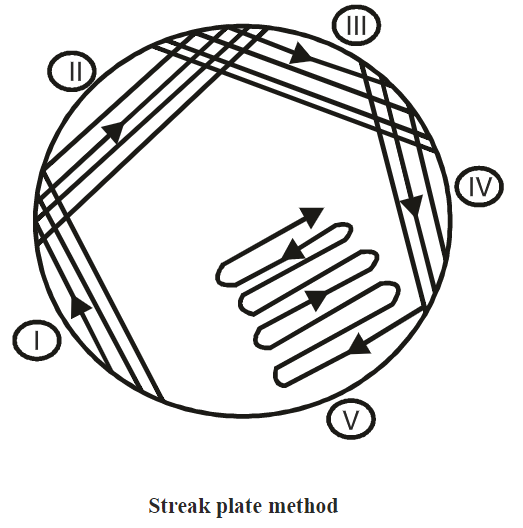
Also Read: Streak plate method: Principle, Procedure, Results
Lawn Culture or Carpet Culture
Lawn cultures are prepared by flooding the surface of the plate with a liquid culture or suspension of the bacterium, pipetting off the excess inoculum, and incubating the plate. Alternatively, the surface of the plate may be inoculated by applying a swab soaked in the bacterial culture or suspension. After incubation, lawn culture provides a uniform growth of the bacterium.
Uses
- Antibiotic susceptibility testing: It is useful for antibiotic susceptibility testing by disk diffusion method
- Bacteriophage typing.
Stroke Culture
Stroke culture is made in tubes containing agar slope or slant. Slopes are seeded by lightly smearing the surface of agar with a loop in a zig-zag pattern taking care not to cut the agar. It is employed for providing pure growth of the bacterium for slide agglutination and other diagnostic tests.
Stab Culture
In the preparation of the stab cultures, a suitable medium such as nutrient gelatin or glucose agar is punctured with a long, straight, charged wire into the center of the medium and withdrawing in the same line to avoid splitting the medium. The medium is allowed to set, with the tube in the upright position, providing a flat surface at the top of the medium.
Uses
- Mainly for demonstration of gelatin liquefaction.
- Demonstration of oxygen requirement of the bacterium under study.
- For the maintenance of stock cultures.
- To study the motility of bacteria in semisolid agar.
Pour-Plate Culture
This method is used for counting the number of living bacteria or groups of bacteria in liquid culture or suspension. A measured amount of the suspension is mixed with molten agar medium in a Petri dish. Either 1.0 ml or 0.1 ml of dilutions of the bacterial suspension are introduced into a Petri dish. The nutrient medium, in which the agar is kept liquid by holding it in a water bath at 45-50°C, is poured over the sample, which is then mixed into the medium by gentle agitation of the plate. When the agar solidifies, the plate is incubated inverted at 37°C for 48 hours, or most suitable for the species examined.
After incubation, colonies will grow within the nutrient agar (from cells suspended in the nutrient medium as the agar solidifies) as well as on the surface of the agar plate and can be enumerated using colony counters
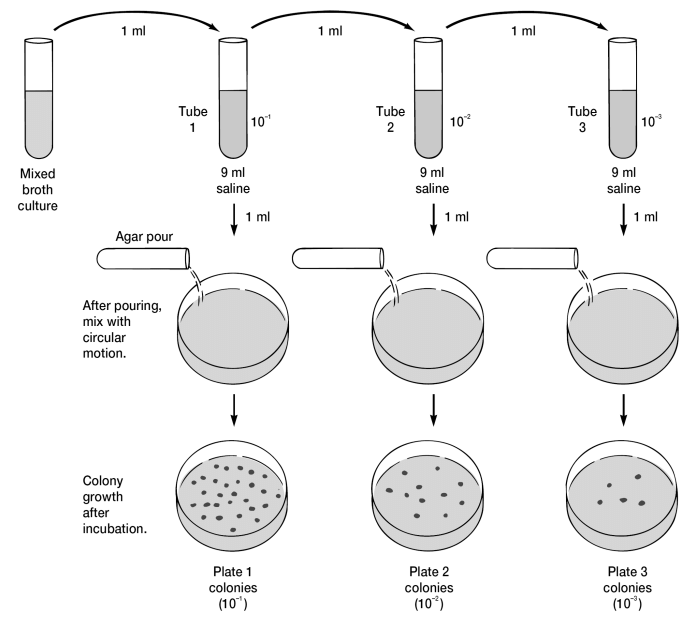
Uses
- Gives an estimate of the viable bacterial count in a suspension.
- The recommended method for quantitative urine cultures.
Also Read: Pour Plate Technique: Principle, Procedure, Results
Shake Culture
It is made by melting nutrient agar in a test tube, cooling it to 45°C, and inoculating it while molten from a liquid medium with a drop from a capillary pipette or a wetted straight wire, depending on the desired size of the inoculum. Withdraw the pipette or wire and flame the mouth of the tube if it has a cotton-wool plug. Replace the cap or plug and discard the pipette into disinfectant or flame the wire. Mix the contents of the tube by rotation between the palms of the hands before the agar solidifies. Incubate it at 37°C for 24 hours and look for the growth of the organisms.
Liquid Cultures
Liquid cultures in tubes, bottles, or flasks may be inoculated by touching with a charged loop or by adding the inoculum with pipettes or syringes.
Uses
- Blood culture and for sterility: Large inocula can be employed and hence this method is adopted for blood culture and for sterility tests, where the concentration of bacteria in the inocula is expected to be small.
- Dilution in the medium For inocula containing antibiotics and other inhibitory substances liquid cultures are preferable, as these are rendered ineffective by dilution in the medium.
- Large yields: Liquid cultures are also preferred when large yields are desired, the yield being enhanced by agitation, aeration, the addition of nutrients, and removal of toxic metabolites (continuous culture methods).
Disadvantages
- It does not provide a pure culture from mixed inocula
- Identification of bacteria is not possible
