QUANTIFYING HETEROTROPHIC MICROBES USING THE SPREAD-PLATE TECHNIQUE
Introduction
Heterotrophs are the community of (yeast, molds, and bacteria) micro-organisms that use organic carbon as feed and are present in any form of water, in comparison to autotrophic ones such as algae that use the sunlight. Chemical compounds are the basis of nutrition for heterotrophic bacteria.
Types-
- Parasitic
- Saprophytic
- Symbiotic
A large measure for the analysis of water is the quantification of heterotrophic bacteria. In municipal water systems (MWS) there are several factors that determine the microbiological quality of drinking water. Free chlorine and/or chloramines are typically used for minimizing bacterial recovery and/or regeneration in WDS. Despite these preventive measures, the growth in bulk water and biofilms still remains completely under the control of heterotrophic and opportunistic bacteria.
Now, we will discuss the quantification of heterotrophic bacteria using the spread plate technique.
Theory
The diluted sample is pipetted onto the surface of a solidified agar medium and spread with a sterilized, bent glass rod (glass spreader) for the determination of heterotrophic plate count using the spread–plate method.
Objective of study, test organisms:
Environmental sample (e.g. soil or water)
Materials and equipment:
- Agar plates
- Glass spreader (alcohol for sterilization)
- Pipettes, sterile pipette tips
- 99 mL sterile distilled water in a flask
- 9 mL sterile distilled water in test tubes
- Vortex mixer
- Bunsen burner
- Incubator
Procedure
1. Make a 10-fold dilution series from an environmental sample: measure 1 g soil sample into a flask containing 99 mL sterile water (or 1 mL water sample to 99 mL sterile water) mix thoroughly with a vortex mixer, pipette 1 mL from this suspension into a test tube containing 9 mL sterile water, mix thoroughly with vortex, pipette 1 mL from this latter suspension into another test tube containing 9 mL sterile water, mix thoroughly, etc. (until the desired degree of dilution is reached). Use unambiguous labeling throughout the practice (indicate sample name, degree of dilution, etc.).
2. Spread 0.1 mL from the given dilution onto the surface of agar plates: pipette 0.1 mL from the appropriate member of the dilution series onto the center of the agar surface; rinse the glass spreader with alcohol (remove any excess alcohol) and sterilize the rod by flaming (take the rod away from the flame while the alcohol burns); cool down the glass spreader by touching the medium surface (without touching the liquid containing bacteria); spread the liquid evenly over the surface (while spreading dish should be opened only slit-like).
3. Incubate Petri dishes at 28°C for one week.
4. Count the number of discrete colonies, in the case of parallel plates, average the numbers and calculate the CFU value of the sample. Results of different dilutions should also be averaged. Give the CFU values of the original sample in CFU/mL or CFU/g units.
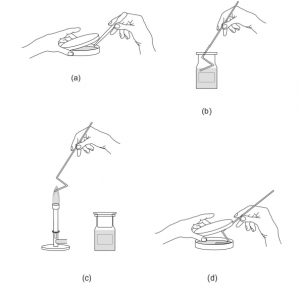
(d) Spread the sample evenly over the surface of agar using the sterile glass spreader, carefully rotating the Petri dish underneath at the same time.
References
https://www.h2odistributors.com/pages/contaminants/contaminant-heterotroph.asp#:~:text=Heterotrophs%20are%20a%20group%20of,Heterotrophic%20Plate%20Count%20(HPC).https://www.researchgate.net/publication/51791899_Heterotrophic_bacteria_in_drinking_water_distribution_system_A_reviewhttps://pubmed.ncbi.nlm.nih.gov/20835882/#:~:text=Quantification%20of%20heterotrophic%20bacteria%20is,quality%20criteria%20and%20detection%20limits.
