(Greek Word: auxein – to grow)
Auxin is defined as an important plant growth regulator or plant hormone which is responsible for terminal elongation of shoot.
Discovery:
Darwin in 1880 noticed the presence of growth regulating substance at the tip of canary grass (Phalaris canariensis).
Boyson-Jensen in 1913 worked on decapitated coleoptile tip.
F.W Went in 1926 worked on oat (Avena sativa) coleoptile tip and identified a substance which caused the curvature of tip towards the light (phototropism). He used the term Indole acetic acid (IAA) for such substance. It is called Avena curvature test for the Bioassay of auxin.
The organic substances which function like IAA are known as auxins. The term auxin was given by Kogl and Haagen Smit (1933).
Kogl and Haggen Smit (1931) obtained auxin a (Auxin triolic acid – C18H22O5) from human urine, auxin b (auxinolonic acid – C18H22O4) from corn germ oil and heteroauxin from human urine. Actually heteroauxin is true auxin or IAA.
Plants also contain some other compounds which cause the same response as IAA and considered to be auxins (Wightman and Lighty, 1982).
Distribution: Auxins are distributed throughout the pant body especially in growing points (apical and other buds).
Translocation: It may be downward (through phloem) or upward (through xylem). Recently it has been found that it is polar through medullary rays. Rubery and Sheidrake (1973 and 1974) and Raven (1975) have been a theory of Chemiosmotic polar diffusion of auxin.
Degradation: Auxin is degraded by two methods: Dark enzymatic and Photo-oxidation.
Solubility: IAA is soluble in organic solvent like chloroform, ether, ethyl alcohol etc.
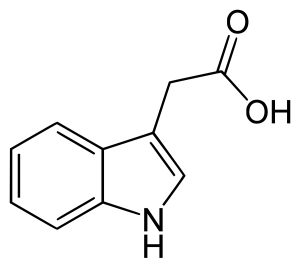
Types of Auxins: Two types:
Natural auxins:
- Indole-3-acetic acid.
- Indole-3-pyruvic acid.
- Indole-3-acetaldehyde.
- Indole-3-ethanol.
Synthetic auxins:
- Indole butyric acid.
- Indole propionic acid.
- 2,4-D (2,4-Dichlorophenoxy acetic acid).
- 2,4, 5-T (2,4,5-Trichlorophenoxy acetic acid).
- Alpha naphthalene acetic acid.
Anti-auxins: 2,4-D, 2,4,5-T and 2,4,5-TIBA are anti-auxins which prevent the action of auxins.
Biosynthesis: Auxins are similar to amino acid Tryptophan.
Role of auxins:
1.Apical dominance: Auxins (IAA and IBA) are responsible for apical dominance. Apical dominance is a phenomenon by which the apical bud inhibits the growth of lateral buds and lateral shoots. Lateral buds are more sensitive to higher concentration than the apical buds. It is known as ‘Theory of Direct inhibition by auxin’. Auxins are present in apical buds. The plants with strong apical dominance grow tall and become unbranched. Maximum apical dominance is seen in palm, datepalm, coconut etc. Formation of lateral buds in potato tubers can be stopped upto three years by spraying them with IAA.
2.Growth: Auxins promote growth of plants. They promote shoot growth when present in high concentration and promote root growth when present in low concentration.
3.Cell enlargement: It is the most important role of auxins. It is due to:
- Loosening of cell wall microfibrils.
- Synthesis of more wall materials.
- Mobilization of carbohydrates.
- Increased membrane permeability and
- Development of higher osmotic pressure.
4.Germinations: Few auxins like IAA, IBA NAA etc. are used to promote seeds germinations.
5.Rootings: Auxins cause the formation of new root initials. In rose, Boungainvillea etc. large number of roots develops in a cut ends when placed in dilute solution of auxins. Recent works have shown that conjugated auxins are more effective than free forms in inducing rooting. e.g., IBA-alanine, IBA-phenylalanine (Felker and Clark, 1981).
6.Cell division in cambium: Auxins induce cell divisions in cambia. Hence auxins are used in tissue culture for the formation of callus. Stimulation of cambial activities in trees by exogenous application of auxins was first reported by Soding.
7.Flowering: Auxins like IAA, IBA, 2,4-D etc. inhibit flowering but in pineapple they promote flowering.
8.Opening of flower buds: Synthetic auxins are used for this purpose.
9.Formation of parthenocarpic fruits: Few auxins like IAA, IBA NAA etc. are responsible for this purpose in cucumber, watermelon, cucurbits, tomato, apple etc. Natural parthenocarpy is found in pineapple, banana etc. because their ovaries are rich in natural auxins.
10.Sweetening of fruits: Sugarcane accumulate more sugars when supplied with IBA, 2,4-D or maleic hydrazide etc. Other fruits can become sweeter with the similar application because these auxins help in conversion of starch into sugar.
11.Frost resistance: Frost resistance with the growth of fruits in many trees 2,4,5-T and sodium salt of NAA also help in the production of normal fruits in the presence of frost.
12.Prevention of pre-mature falling of leaves and fruits: Auxins are used for these purposes because they prevent the formation of abscission layer in apple, pear, orange, cabbage, cauliflower etc. Leaves are shed (fall) seasonally because they stop producing auxins.
13.Prevention of lodging: Spray of auxins prevents the lodging (bending) of legumes, cereals etc.
14.Healing of wounds: Traumatic acid a type of auxin produced in injured part causes healing of wounds by increasing the rate of cell divisions.
15.Weedicides: Few synthetic auxins like 2,4-D, 2,4,5-T etc. are used as weedicides. 2,4-D kills small dicot weed plants.
16.Dormancy: Few auxins like NAA and their derivatives are used to prolong the dormancy of buds in potato.
17.Breaking of Dormancy: IAA, IBA etc. are used to break dormancy in corms, tubers, bulbs, seeds etc.
18.Overcoming sterility: In certain cases NAA (0.001%) has been used to overcome sterility because it allows the pollen grains to germinate rapidly. Side by side it prevents the abscission of ovary.
19.Shortening of internodes: Auxins are used for this purpose. It is generally observed in plants like apple and pear where both types of branches like short and long are found and fruits are borne only on short branches.
20.Respiration: Auxins stimulate respiration (Banner, 1933) by increasing the supply of ADP for the formation of ATP.
