Types of Fermentation
#1. Batch fermentation
- A tank of fermentors is filled with the prepared mash of raw material to be fermented. The temperature and pH for microbiafil fermentation are properly adjusted, and occasionally nutritive supplements are added to the prepared mash.
- The mash is steam sterilized in a pure culture process. The inoculum of a pure culture is added to the fermentor from a separate pure culture vessel.
- Fermentation proceeds and after the proper time the contents of the fermentor are taken out for further processing.
- The fermentor is cleaned and the process is repeated. Thus, each fermentation is a discontinuous process divided into batches.
#2. Continuous fermentation
- Continuous fermentations are those in which fresh nutrient medium is added either continuously or intermittently to the fermentation vessel, accompanied by a corresponding continuous or intermittent withdrawal of a portion of medium for recovery of cells or fermentation product.
- In continuous fermentation, the substrate is added to the fermentor continuously at a fixed rate. This maintains the organisms in the logarithmic growth phase. The fermentation products are taken out continuously.
- But the design and arrangements for continuous fermentation are somewhat complex than batch fermentation.
- Whereas in batch fermentation large volume of nutrient medium is inoculated and growth and biochemical synthesis are allowed to proceed only until maximum yields have been obtained. This is because of the limitation of one or more of the essential nutrients.
- At this point, the batch fermentation is stopped for product recovery.
Methods of continuous fermentation – Continuous fermentation can be conducted in various ways. It can be carried out as-
i) Single-stage fermentation – In this type of fermentation, the fermentor is inoculated and then kept in continuous operation by balancing the input and output of nutrient solution and harvested culture, respectively.
ii) Recycle fermentation – In recycled continuous fermentation, a portion of the withdrawn culture or of unused residual substrate plus the withdrawn culture is recycled to the fermentation vessel.
iii) Multistage fermentation –Multiple stages continuous fermentation involves two or more stages with the fermentors which are operated in sequence. The fermentation is divided into two phases, i) growth phase occurs in the first fermentor followed by ii) synthesis phase in the second fermentor. The multiple-stage continuous fermentation is particularly applicable to those fermentations in which the growth and synthetic activities of the cells are not simultaneous i.e. product formation occurs only after the cell multiplication rate has slowed.
- There are different ways in which microbial activity in continuous culture can be controlled.
- This includes a chemostat and turbidostat. In both approaches, it is necessary to maintain a constant cell population in fermentors or fermentors.
- In this connection, the feed of fresh nutrients is critical as it is related to the generation time of the organism.
- The very low flow rate of feed can allow the culture to go into a maximum stationary phase of growth and thus the continuous aspect of fermentation will not be maintained.
- Whereas very high flow rate can dilute the cell population that and thus the continuous aspect of fermentation will not be maintained.
Advantages
1) The productivity of continuous fermentation is greater than that of batch fermentation.
2) Continuous fermentation appears to be better because of two factors because the fermentation equipment is in constant usage with little shutdown time and after initial inoculation, further production of inoculum is not required.
Disadvantages
1) It requires thorough knowledge of the dynamic aspects of microbial behavior and growth which is deficient in most of the industrial fermentation processes because of the complexities of the growth and synthetic pattern of the organism.
2) Contamination and mutation in producing strain are the distinct problems for the development of a successful continuous fermentation process.
3) Continuous fermentation often wastes nutrient substrate. A fermentation broth is continuously withdrawn for product recovery contains a certain amount of the residual unused nutrients of the medium as well as a portion of the fresh medium which is being continuously added.
4) In a process that employs the use of viscous media, adequate mixing of nutrients and inoculum is not possible.
5) Continuous fermentations become more complex and difficult to accomplish when a chemical product and not a microbial cell is the desired product. This is because the conditions which are optimal for growth are not optimal for the formation of a chemical fermentation product.
Example – Antibiotic fermentations.
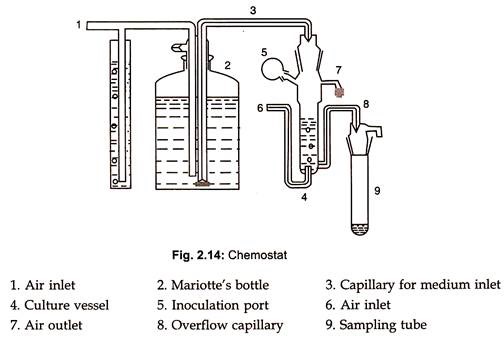
i) Chemostat
- Chemostat maintains the nutrient feed and harvest culture withdrawal rates at constant values, but always less than that which allows a maximum growth rate.
- The growth rate is controlled by supplying only a limiting amount of critical growth factor in the feed solution.
- Thus, cell multiplication cannot proceed at a rate greater than that allowed by the availability of this critical nutrient.
- The controlling factor for growth can be a high concentration of the toxic product of the fermentation and the pH value of the temperature of incubation.
Advantages
The chemostat operation of continuous fermentation is more often used than turbidostat operation because.
1) Fewer mechanical problems are encountered.
2) Occurrence of less residual unused nutrient in the harvested culture.
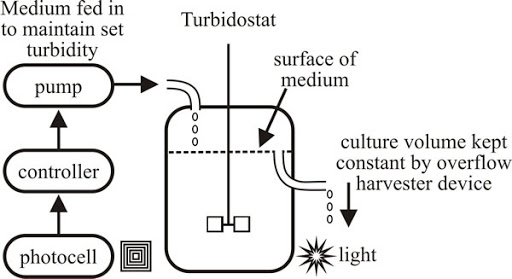
ii) Turbidostat
- In the turbidostat, the total cell population is held constant by employing a device that measures the culture turbidity so as to regulate both nutrient feed rate to the fermentor and the culture withdrawal rate from the fermentor.
- If a population rate rises above a predetermined level, a greater amount of fresh medium is added to the fermentor so as to dilute the cell concentration.
- Thus, there is no limiting nutrient consciously imposed with this process so that the cell growth rate should always be maximal and the growth is maintained in the logarithmic phase.
Disadvantages
1) Since the culture is to be maintained in the logarithmic phase, the fermentation is operated at lower cell concentration and thus may require a longer period.
2) This causes a greater loss of residual unused nutrients with the withdrawn harvested culture.
Applications of continuous culture
1) Turbidostat provides a constant source of cells in an exponential phase of growth, and they allow cultures to be grown continuously at extremely low concentrations of substrate.
2) Growth at low substrate concentrations is valuable in studies on the regulation of synthesis or catabolism of the limiting substrate, in the selection of various classes of mutants, and in ecological studies.
#3. Fed-batch fermentation
Two basic approaches to the fed-batch fermentation can be used: the constant volume fed-batch culture – i) Fixed Volume Fed-Batch – and the ii) Variable Volume Fed-Batch.
i) Fixed volume fed-batch
- In this type of fed-batch, the limiting substrate is fed without diluting the culture. The culture volume can also be maintained practically constant by feeding the growth limiting substrate in undiluted form, for example, as a very concentrated liquid or gas (ex. oxygen).
- Alternatively, the substrate can be added by dialysis or, in a photosynthetic culture. Radiation can be the growth limiting factor without affecting the culture volume.
- A certain type of extended fed-batch-the cyclic fed-batch culture for fixed volume systems – refers to a periodic withdrawal of a portion of the culture and use of the residual culture as the starting point for a further fed-batch process.
- Basically, once the fermentation reaches a certain stage, (for example, when aerobic conditions cannot be maintained anymore) the culture is removed and the biomass is diluted to the original volume with sterile water or medium containing the feed substrate.
- The dilution decreases the biomass concentration and result in an increase in the specific growth rate.
- Subsequently, as feeding continues, the growth rate will decline gradually as biomass increases and approaches the maximum sustainable in the vessel once more, at which point the culture may be diluted again.
ii) Variable volume fed-batch
- As the name implies, a variable volume fed-batch is one in which the volume changes with the fermentation time due to the substrate feed.
- In this type of fermentation, once the fermentation reached a certain stage after which is not effective anymore, a quantity of culture is removed from the vessel and replaced by a fresh nutrient medium.
- The decrease in volume results in an increase in the specific growth rate, followed by a gradual decrease as the quasi-steady-state is established.
- As the name implies, a variable volume fed-batch is one in which the volume changes with the fermentation time due to the substrate feed.
- In this type of fermentation, once the fermentation reached a certain stage after which is not effective anymore, a quantity of culture is removed from the vessel and replaced by a fresh nutrient medium.
- The decrease in volume results in an increase in the specific growth rate, followed by a gradual decrease as the quasi-steady-state is established.
Single fed-batch process
This type refers to a type of fed-batch in which supplementary growth medium is added during the fermentation, but no culture is removed until the end of the batch. This system presents a disadvantage over the fixed volume fed-batch and the repeated fed-batch process: much of the fermentor volume is not utilized until the end of the batch and consequently, the duration of the batch is limited by the fermentor volume.
Advantages of the fed-batch reactors
i) Under controllable conditions and with the required knowledge of the microorganism involved in the fermentation, the feed of the required components for growth and/or other substrates required for the production of the product can never be depleted and the nutritional environment can be maintained approximately constant during the course of the batch.
ii) The production of by-products that are generally related to the presence of high concentrations of substrate can also be avoided by limiting its quantity to the amounts that are required solely for the production of the biochemical.
iii) Sometimes, controlling the substrate is also important due to catabolic repression. Since fed-batch method usually permits the extension of the operating time, high cell concentrations can be achieved and thereby. improved productivity [mass of product/(volume.time)]. This aspect is greatly favored in the production of growth-associated products.
iv) This method allows the replacement of water loss by evaporation and decrease of the viscosity of the broth such as in the production of dextran and xanthan gum, by addition of a water-based feed.
v) Fed-batch process is ideal in fermentations dealing with toxic or low solubility substrates.
vi) In the case of recombinant strains, the fed-batch mode can guarantee the presence of an antibiotic throughout the course of the fermentation. Since the growth can be regulated by the feed and knowing that in many cases a high growth rate can decrease the expression of encoded products in recombinant products, the possibility of having different feeds and feed modes makes fed-batch an extremely flexible tool for control in these cases.
vii) Because the feed can also be multi-substrate, the fermentation environment can still be provided with required protease inhibitors that might degrade the product of interest, metabolites, and precursors that increase the productivity of the fermentation.
viii) In fed-batch fermentation, no special equipment is required in addition to that one required by a batch fermentation, even considering the operating procedures for sterilization and the preventing of contamination.
Disadvantages of the fed-batch reactors
i) Fed-batch fermentation is a production technique in between batch and continuous fermentation. A proper feed rate, with the right component constitution, is required during the process.
ii) When high concentrations of a substrate are present. the cells get overloaded, this is. The oxidative capacity of the cells is exceeded, and due to the Crabtree effect, products other than the one of interest are produced, reducing the efficacy of the carbon flux. Moreover, these by-products prove to even contaminate the product of interest, such as ethanol production in baker’s yeast production, and to impair the cell growth reducing the fermentation time and its related productivity.
Advantages of cyclic fed-batch
i) Production of high cell densities due to an extension of working time (particularly important in the production of growth-associated products)
ii) Controlled conditions in the provision of substrates during the fermentation, particularly regarding the concentration of specific substrates as for example the carbon source control over the production of by-products or catabolite repression effects due to limited provision of substrates solely required for product formation the mode of operation can overcome and control deviations in the organism’s growth pattern as found in batch fermentation
ii) It allows the replacement of water loss by evaporation
iv) Alternative mode of operation for fermentations leading with toxic substrates (cells can only metabolize a certain quantity at a time) or low solubility compounds increase of antibiotic- marked plasmid stability by providing the correspondent antibiotic during the time span of the fermentation.
v) No additional special equipment is required as compared with the batch fermentation mode of operation.
Disadvantages of cyclic fed-batch
i) It requires previous analysis of the microorganism, its requirements and the understanding of its physiology with the productivity
ii) It requires a substantial amount of operator skill for the set-up, definition and development of the process
iii) In a cyclic fed-batch culture, care should be taken in the design of the process to ensure that toxins do not accumulate to inhibitory levels and that nutrients other than those incorporated into the feed medium become limiting, Also, if many cycles are run, the accumulation of non-producing or low-producing variants may result.
iv) The quantities of the components to control must be above the detection limits of the available measuring equipment.
#4. Anaerobic Fermentation:
A fermentation process carried out in the absence of oxygen is called anaerobic fermentation. There are two types of anaerobic microorganisms viz, obligate anaerobic microorganisms and facultative anaerobic microorganisms. The former like Clostridium sp. cannot withstand oxygen or remain active only in the absence of oxygen.
They remain active in the absence of oxygen and produce the optimum amount of the desired product. The facultative anaerobes like lactic acid bacteria are able to withstand a small amount of oxygen. However, certain organisms like yeast require initial aeration to build up a high cell yield before anaerobic conditions are created.
Anaerobic conditions in the fermenter are created either by withdrawing the oxygen present in the headspace by an exhaust pump and pumping some inert gases like nitrogen, argon, etc., or by flushing it out, by the emergence of certain gases like carbon dioxide or hydrogen.
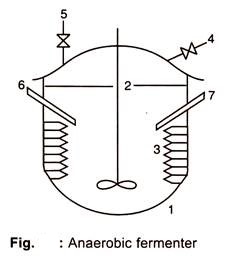
Stationary medium and viscous medium also create anaerobic conditions. Sometimes in order to create an anaerobic condition, the medium is inoculated at the bottom of the fermenter soon after sterilization.
(a) Merits:
1. Production of economically valuable byproducts like carbon dioxide and hydrogen gas during anaerobic fermentation, which may fetch some profits to the manufacturers.
(b) Demerits:
1. Manufacturers may have to spend more money in providing extra provisions to the fermenter like an exhaust pump in order to enforce anaerobic conditions.
2. It requires special media like viscous media whose preparation requires certain costly chemicals.
#5. Aerobic Fermentation:
A fermentation process carried out in the presence of oxygen is called aerobic fermentation. In most of the commercial processes and the majority of the products of human utility are produced by this type of fermentation.
Fermentation can be surface culture or static and submerged.
#6. Surface Fermentations:
Surface fermentations are those where the substratum may be solid or liquid. The organism grows on the substratum and draws the nutrients from the substratum. These types of fermentations are desirable where the products are based on sporulation. But it has several disadvantages such as it exposes the organism to unequal conditions, both oxygen, and nutrients.
#7. Submerged Fermentations:
Submerged Fermentations are those in which the nutrient substratum is liquid and the organism grows inside the substratum. The culture conditions are made uniform with the help of spargers and impeller blades. Most of the industrial fermentations are of this type. The substratum which is in a liquid state and such medium is also called broth.
#8. Solid Substrate/State Fermentation:
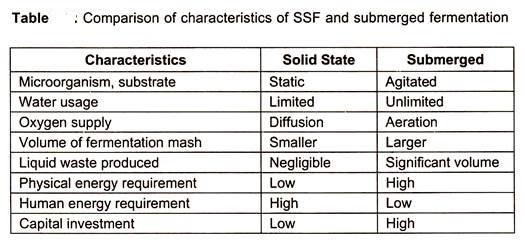
Solid-state (substratum) fermentation (SSF) is generally defined as the growth of the microorganism on moist solid materials in the absence or near the absence of free water. In recent years SSF has shown much promise in the development of several bioprocesses and products, SSF has been ambiguously used as solid-state fermentation or solid-substrate fermentation.
However, it is proper to distinguish between two processes. Solid substrate fermentation should be used to define only those processes in which the substrate itself acts as a carbon source occurring in the absence or near absence of free water. On the other hand, solid-state fermentation is that fermentation that employs a natural substrate as above or an inert substrate used as solid support. Solid substrate fermentation is normally many step process involved.
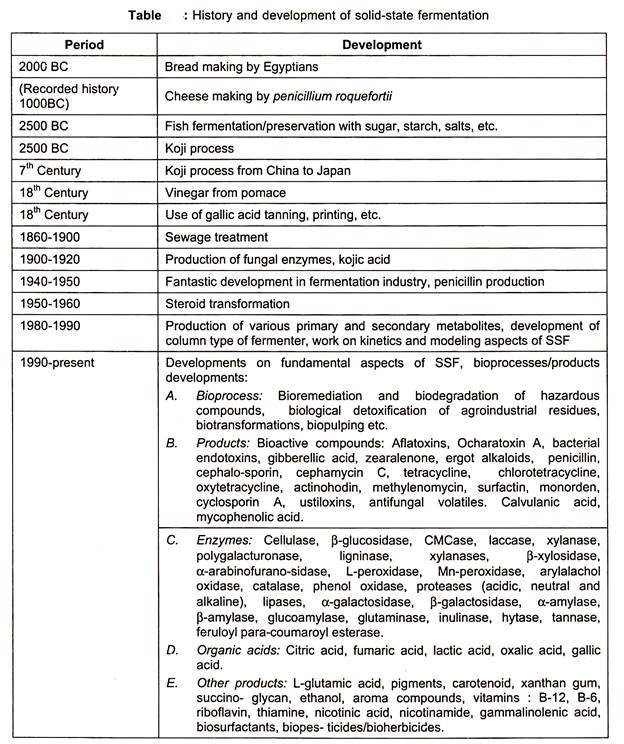
SSF has a long history and some of the main events are précised in the table below.
A comparison of solid-state and submerged fermentation is given in the table below.
Based on the need for aeration and agitation, SSF can be divided into two groups:
(a) Fermentation without agitation.
(b) Fermentation with occasional or continuous agitation.
The second group can be further divided into:
(i) Fermentation with occasional agitation, without forced aeration.
(ii) Fermentation with slow continuous agitation with forced agitation.
(iii) Pretreatment of a substratum that often requires either mechanical, chemical or biological processing.
(iv) Hydrolysis of polymeric substrates such as polysaccharides and proteins.
(v) Utilization of hydrolysis products.
(vi) Separation and purification of end products.
(vii) Fermentation with occasional agitation and forced aeration.
(viii) Fermentation with slow continuous agitation and forced aeration.
Several types of fermenters have been used for solid-state fermentation. Laboratory studies have generally been carried out in flasks, beakers, Roux bottles, Petri dishes, glass jars, and columns. Inoculum is added after substrate autoclaving and incubated without any agitation and aeration.
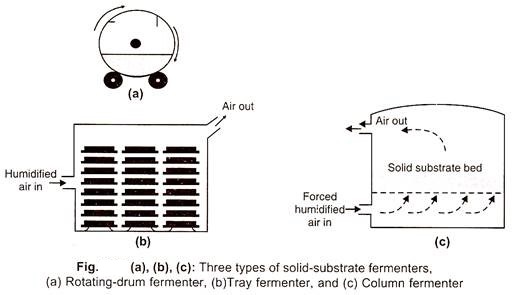
For large-scale SSF bioprocess, three types of fermenters are in operation:
(a) Drum Fermenter:
It basically consists of drum type vessel usually equipped with a rotating device and arrangements for air circulation . The air inlet pipe may run parallel to the bottom or center or it may branch at several points over the whole length of the drum to facilitate air distribution which is normally attained by forced aeration, thus achieving the mixing of the fermenting substratum. Growth of the microorganism in this type of fermenter is considered to be better and more uniform than the tray fermenter.
(b) Tray Fermenter:
Tray fermenters are the simplest and can be constructed using wood, metals, or plastic material. The bottom of the tray is perforated in such a way that it holds the substrate and allows aeration. Kofi fermentation has traditionally been carried out in a tray fermenter. Tray fermenter, however, requires a large operational area and is labor-intensive. Their design does not lead readily to mechanical handling. The substrate requires separate sterilization.
(c) Column Fermenter:
The column fermenter consists of a glass or plastic column with lids at both ends. It may be fitted with a jacket for the circulation of water to control the temperature of fermenting substrate. Alternatively, the whole column may be placed in a temperature-controlled water bath. Usually, the air is circulated from bottom to top.
The column may be vertical or horizontal as per convenience. Bed reactor is simple in design in which humidified air is pumped into substratum and the used waste gases go out through the outlet provided continuous agitation with forced air to prevent adhesion and aggregation of substrate particles. These systems are very useful for biomass production for animal feed.
Microorganisms associated with solid substrate fermentation are those that tolerate relatively low water activity down to 0.7. They may be employed in the form of monocultures as in mushroom production e.g. Agaricus bisporus. Dual cultures e.g. straw conversion using Chaetomium cellulolyticum and Candida tropicalis. Mixed cultures as used in compositing and the preparation of silage where the microorganisms may be indigenous or added as mixed starter cultures.
For some fermentation, SSF is desirable because of the following reasons:
1. In several productions, the product formation has been found superior in the solid culture process.
2. The most commonly used microorganisms in the production of secondary metabolites are fungi and actinomycetes and the mycelial morphology of such organisms is ideal for their invasive growth on solid and insoluble substrates.
3. The fungal morphology is responsible for considerable difficulties in large-scale submerged processes. These include highly viscous non-Newtonian broths and foam production. This results in very high power requirements for mixing and oxygen transfer. The presence of chemical antifoam in fermentation broth reduces oxygen transfer efficiency and can lead to problems in product recovery.
4. In some processes the final product is required in solid forms, such as antibiotics in animal feed.
5. The capital cost of the overall production process is claimed to be significantly less.
6. The yields of certain secondary metabolites such as aflatoxin B1 and ochratoxin A obtained from liquid culture were found to be very poor. This led to the use of SSF to get a higher yield of mycotoxins (100 g).
7. The fungus possesses tremendous turgor pressure at the mycelial tips.
8. Microbial cells attach to solid substrate particles and completely surround the particle in mycelial webs.
9. It provides the optimum quantity of water (aw) for growth.
10. Crude substrates can be used as the organisms can tolerate high concentrations of metal ions and mineral ions.
11. Overcome catabolite repression and can be provided high substrate concentration.
12. Enzymes become extracellular otherwise intracellular in SMF. E.g.- Galactase, tannase and invertase.
13. Metabolite production phase is long.
14. Co-production of carbohydrates and proteases.
15. Enzymes produced by this will be with better properties and extra desirable components.
16. Fermentation of straw eliminates costly centrifugation and dewatering.
17. Lower capital and recurring expenditure.
18. Low wastewater output/less water need.
19. Reduced energy requirement.
20. Absence of foam formation.
21. Simplicity.
22. High reproducibility.
23. Simpler fermentation media.
24. Lesser fermentation space.
25. Absence of rigorous control of fermentation parameters.
26. Easier aeration.
27. Economical to use even on smaller scales.
28. Easier control of contamination.
29. Applicability of using fermented solids directly.
30. Storage of dried fermented matter.
31. Lower cost of downstream processing
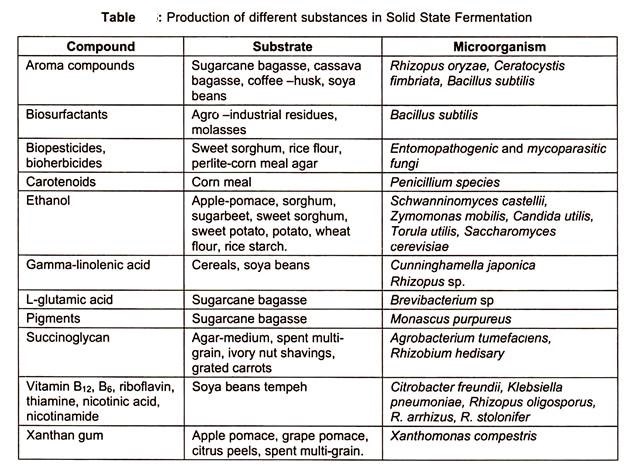
Some of the substances produced by SSF are precise in the table below:
You May Read:
