Strain Improvement Methods
Strain improvement is one of the essential steps in fermentation for obtaining the highest possible yield and is economical. This mainly depends upon the efficiency of the production strain involved in the fermentation process. Active microbial strains obtained by primary and secondary screening are not so efficient as can be used in fermentation processes. Therefore, such strains require improvement for production of high yield compounds. Main objectives for strain improvement are:
(1) To obtain highly stable and infection resistant strains.
(ii) To produce high yield strains that make the process more economical.
(iii) To obtain a strain which is resistant to medium constituents and can tolerate low oxygen tension.
(iv) To obtain a morphologically favourable strain.
(v) Easy removal of all undesirable constituents from the bioactive strain. Microbial strain improvement can be achieved by mutation and DNA transfer techniques.
Mutation: Microbial cells change their own characteristics by natural cell division. These strains are called mutants. The factors which are responsible for the changes are called mutagens and the process is called a mutation. Mutagenic agents are UV-radiation, X-rays, gamma rays, cobalt 60, nitrogen mustard, ethidium bromide, 5-bromouracil, nalidixic acid etc. General protocol for mutation techniques is given below. These mutants are mainly grouped into two categories such as auxotrophic mutants and mutants resistant to analogues.
Isolation of strains from natural sources
↓
Primary and secondary screening for bioactive strains
↓
Select different types of bioactive strains
↓
Mutagen treatment (UV, X-rays, 60CO, NTG etc.).
↓
Check the activity
↓
Select higher yielding strains
↓
Large-scale testing
↓
Highest yielding strain
↓
Production
Fig. 2.8: General method for mutation
Microorganisms cannot synthesize excess metabolites over limiting the cell’s requirements because cells have some regulatory mechanisms that control the synthesis of metabolites. Suppression of these regulatory mechanisms is necessary to develop the strains to produce higher yields of the required metabolites. Specific mutants are developed which have lost the ability to synthesize one of the end product capable of feedback inhibition or feedback repression. Feedback inhibition means over accumulation of the end product which inhibits the activity or strength of the enzyme catalysing the reaction. Feedback repression means over accumulation of the end product which inhibits the synthesis of the enzyme catalysing the reaction. This may be explained by considering a situation where two end products (X and Y) are produced by branched biosynthetic pathway from substrate ‘A’ as shown in Figure 2.9.
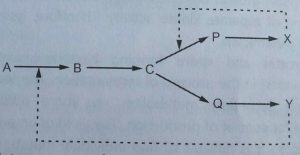
The product (primary metabolite) can show a feedback inhibition on its own production by inhibition of the enzyme catalysing the reaction for formation of ‘B” from ‘A. Product X can show an inhibition of the enzyme catalysing the reaction ‘C’ and P.
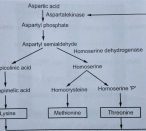
Corynebacterium glutamicum and Brevibacterium flavum produces an important amino acid (lysine) as a primary metabolite. These organisms also produce some other amino-acids like threonine and methionine., The pathway (Fig. 2.10) is subject to feedback inhibition by a mixture of lysine and threonine controlling the activity of aspartate kinase. When these two amino acids are over accumulated as an end product then their synthesis is inhibited by inhibiting enzyme activity. This type of control is termed an ‘absolute feedback inhibition. Isolation of specific mutants can help for avoiding the inhibition of the enzyme activity. This isolated mutant cannot catalyse the conversion of aspartic semialdehyde to homoserine and therefore, prevents the formation of threonine and methionine. A mutant is able to grow in homoserine supplemented medium and the organism is called ‘homoserine auxotroph’. This nutritionally deficient mutant is cultured in a medium with low concentration of threonine and methionine to support growth but not enough threonine concentration to co-operate with lysine in inhibition of asparate kinase activity. Therefore, lysine production increases from Corynebacterium glutamicum.
Moulds, actinomycetes and spore-forming bacteria mainly produce secondary metabolites (e.g. antibiotics) in the process of fermentation. The techniques for isolation of mutants overproducing secondary metabolites are more difficult because very less information is available for control of production. The yield of an antibiotic also depends on various types of genes, it is difficult to find individual mutations that can increase the yield from bioactive strains. The media composition and environmental conditions also strongly influence the production of antibiotics. Various genetically improved strains have been developed through mutation to increase yields . The isolated culture is treated with a mutagen and then mutant colonies are tested for activity.
Spore suspension of isolate (eg. Aenicillium chrysogenium)
↓ Mutagenic treatment
↓ (UV, X-rays NTG,60Co)
Survivors of mutation grown on agar medium
↓
Select the specific colonies (150 to 200 colonies) and suspend separately in saline water
↓
Each cultures are again inoculated into liquid medium for inoculum development
↓
Each inoculum is inoculated into production medium
↓
Best antibiotic producers are identified (10 to 12 colonies) retested, maintained as the stock cultures and remutated
DNA transfer techniques:
Different DNA transfer techniques are used for obtaining high-yielding strains. These techniques are performed by transferring all or part of the DNA to the recipient microbial cell from the donor cell (For details, please refer to this article). Bacterial recombination occurs whenever new gene arrangements are formed through the exchange, elimination or insertion of DNA Recombination has been applied for the production of various industrial strains.
The techniques used for the transfer of genetic material are listed as follows:
- Transformation
- Transduction
- Conjugation
- Parasexuality
- Protoplast fusion
