Micro-RNA (miRNA): Introduction
- miRNA are small, evolutionary, single-stranded, non-coding RNA molecules that bind mRNA targets to inhibit the development of proteins. A variety of regulatory, non-coding RNAs exist in addition to messenger RNA (mRNA), ribosomal RNA(rRNA), and transfer RNA (tRNA), which play central roles within cells (ncRNAs).
- Such non-coding RNAs are of variable length and do not have a long open frame of reading. They do not code proteins, but they do serve as regulators of ribo, and Post-transcriptional modulation of gene expression is their key role.
- Both in prokaryotes and eukaryotes, several ncRNAs have been identified and characterized and are involved in the basic identification of cellular nucleic acid targets by complementary base pairing, cell growth regulation, and differentiation. RNA interference is considered the mechanism of gene control by RNAs (RNAi).
- MicroRNA (miRNA) and small interfering RNA (siRNA) are two types of small ribonucleic acid (RNA) molecules that are essential to RNA interference.
- These small RNAs may bind to other unique molecules of mRNA and either increase or decrease their activity by blocking a protein from being formed by mRNA, for example. In protecting cells against parasitic nucleotide sequences-viruses and transposons, RNA interference has a significant role. It also forms development.
- RNA interference (RNAi) is expressed in many ways: by inhibiting mRNA translation, by killing mRNA, or by transcriptional silencing of the promoter directing the mRNA expression. In the worm Caenorhabditiselegans and the plant Arabidopsis, the functions of these RNAs vary from developmental regulation to mechanisms that shield species against viral infections.
Biogenesis of miRNA
- miRNA is a small non-coding RNA molecule that functions in RNA silencing and post-transcriptional control of gene expression present in plants, animals, and some viruses. Typically, it is 21-22 nucleotides long. microRNA genes are predominately found in intergenic areas. They have also been identified, however in protein-coding gene introns and exons.
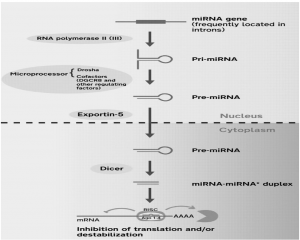
- RNA Polymerase II transcribes miRNA genes. The polymerase binds to a promoter near the sequence of DNA that encodes the primary RNA hairpin loop (pri-miRNA).
- The post-transcriptional modification takes place in the resulting transcript: capping and polyadenylation and splicing. Two RNA cleavage reactions from this long RNA transcript, pri-mRNA, which has a hairpin-shaped secondary structure, produce the short RNAs.
- The stem-loop, called the pre-mRNA, is released by the first cleavage; the second produces from the pre-mRNA the mature miRNA. Two separate RNases mediate the two cleavage reactions required to produce the miRNA from these primary transcripts.
- Dicer is one and Drosha is the other. Two-step nucleolytic processing to produce an active mi RNA: To process the initial primi RNA transcript containing the stem-loop structure into the mature miRNA, two specialized RNA cleaving enzymes are necessary.
- The first enzyme is Drosha, a member of an enzyme family called RNAse III. Drosha creates two cleavages that cut the RNA (pre-miRNA) stem-loop region out of the main transcript RNA (pri-miRNA).
- This enzyme acts along with an essential protein subunit (called Pasha in some species and DGCR8 in others and forms an active microprocessor complex between these two proteins.
- The Drosha-generated pre-miRNA is about 65-70 nucleotides long. Drosha resides in the nucleus, so in the nucleus, this step of cleavage takes place.
- In pri-miRNA, the base-paired stem is about 33bp long, and the loop at the top of this stem is variable in size. The stem region consists of two functional segments: the lower stem (11 bp long) and the upper stem (11 bp long) (22 bp long).
- Drosha cleaves 11 bp, that is, between the lower and upper stems of the pri-miRNA, away from the dsRNA-ssRNA junction.
- In a process involving the nucleocytoplasmic shuttler Exportin-5, the pre-miRNA released by Drosha is exported to the cytoplasm where the second RNA cleavage reaction takes place.
- Pre-miRNA hairpins are exported from the nucleus. This protein, a member of the family of karyopherins, recognizes a two-nucleotide overhang left at the 3 ‘end of the pre-miRNA hairpin by the RNase III enzyme Drosha. In the cytoplasm, the pre-miRNA hairpin is cleaved by the RNase III enzyme Dicer.
- This endoribonuclease interacts with the 3′ end of the hairpin and cuts away the loop joining the 3′ and 5 ‘arms, producing an imperfect miRNA:miRNA duplex of about 22 nucleotides in length. Exportin-5-mediated transport to the cytoplasm is energy-dependent, using GTP bound to the Ran protein.
- A 21-25 nucleotide RNA molecule that guides gene expression regulation is generated by the action of Drosha and Dicer. By forming the RNA-induced silencing complex, regulatory RNAs regulate gene expression (RISC).
- To give a “guide RNA” (an active form of miRNA), a component of RISC and a passenger RNA, which is usually discarded, the miRNA is denatured. RISC is a complex of multi-components that includes the RNA guide and a protein called the RNA cleaving enzyme called Argonaute. “Slicer” is often called Argonaute, and “slicing” is called mRNA cleavage.
miRNA Function
- In the regulation of distinct processes in mammals, miRNAs have important functions. In gene regulation, they provide a key and powerful instrument and thus a potential new class of therapeutic targets.
- MiRNAs play an evolutionarily conserved role in animal growth and various physiological functions. In species, miRNAs largely show minimal complementarity with their target mRNAs, but this is still adequate for certain physiological processes to be controlled.
- The miRNA serves as a guide to negatively regulate its expression through base-pairing with target mRNA.
- The extent of complementarity between the guide and the mRNA target decides which mechanism of silencing will be used; target messenger RNA (mRNA) cleavage with resulting degradation or inhibition of translation.
- In specific tumors, miRNA expression profiles are altered, implying that miRNA may be involved in cancer and other disease development.
References
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4504744/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3315687/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3048316/
More To Read…
